FDA is Recommending Pausing the Use of Johnson and Johnson Vaccine
FDA is Recommending Pausing the Use of Johnson and Johnson Vaccine in the U.S.
Source: Tom Williams / Getty
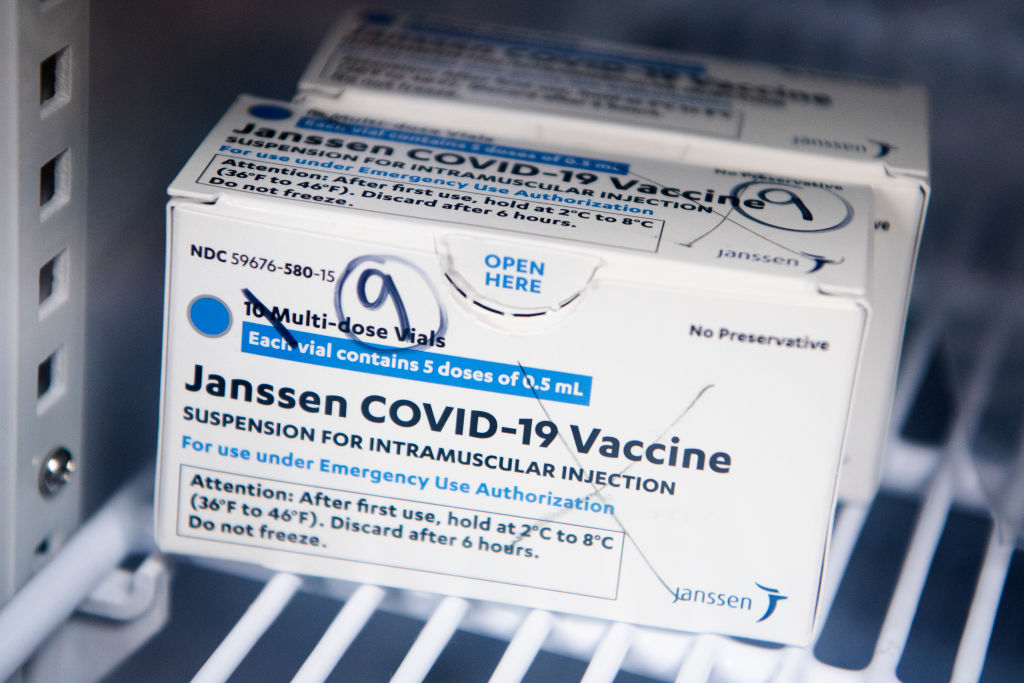
The FDA has announced that the distribution of the Johnson & Johnson COVID-19 will be paused after six women in the U.S. had rare blood-clotting after receiving the vaccine. The women were ranging in ages from 18 to 48, and while six people may not seem like a lot precautions are being taken swiftly. There have been approximately 6.8 million shots given with the majority of people not having issues.
The pausing of the Johnson & Johnson vaccine does not affect the other two vaccines from Pfizer and Moderna. But if you did receive the Johnson & Johnson shot and are having to severe stomach pain, leg cramps, or severe headaches you should call your healthcare provider right away. If you do not know what shot you received, check your CDC vaccination card where it is noted what brand shot you received. If you have an appointment scheduled to receive the Johnson & Johnson COVID-19 vaccine, it is advised to call your provider for further directions.
Get Breaking News & Exclusive Contest in Your Inbox:
[ione_media_gallery id=”3917798″ src=”https://newsone.com/” overlay=”true”]
The Latest:
- OnlyFans Models Go Viral For Cheering On San Antonio Spurs, Thirsty Creeps Pop Out
- Louisiana Passes Map Eliminating Majority-Black District
- Viral Fight Breaks Out During A Virginia High School Graduation
- Op-ED: Why Can’t Our Bigoted President Leave The Somali Community Alone?
- The Playlist: Songs For When You’re Ready To Tell Her How You Feel
- Memphis Residents File Lawsuit Alleging ICE Is Violating Constitutional Rights
- Jaxson Dart Speaks With Teammates Following Donald Trump Intro, Social Media Still Reacting
- Terry Rozier Accused Of Taking $100K Bribe To Leave NBA Game Early
- Coco Gauff’s Most Elite Moments
- Federal Judge Issues Block On Trump’s Anti-Weaponization Fund








