FDA is Recommending Pausing the Use of Johnson and Johnson Vaccine
FDA is Recommending Pausing the Use of Johnson and Johnson Vaccine in the U.S.
Source: Tom Williams / Getty
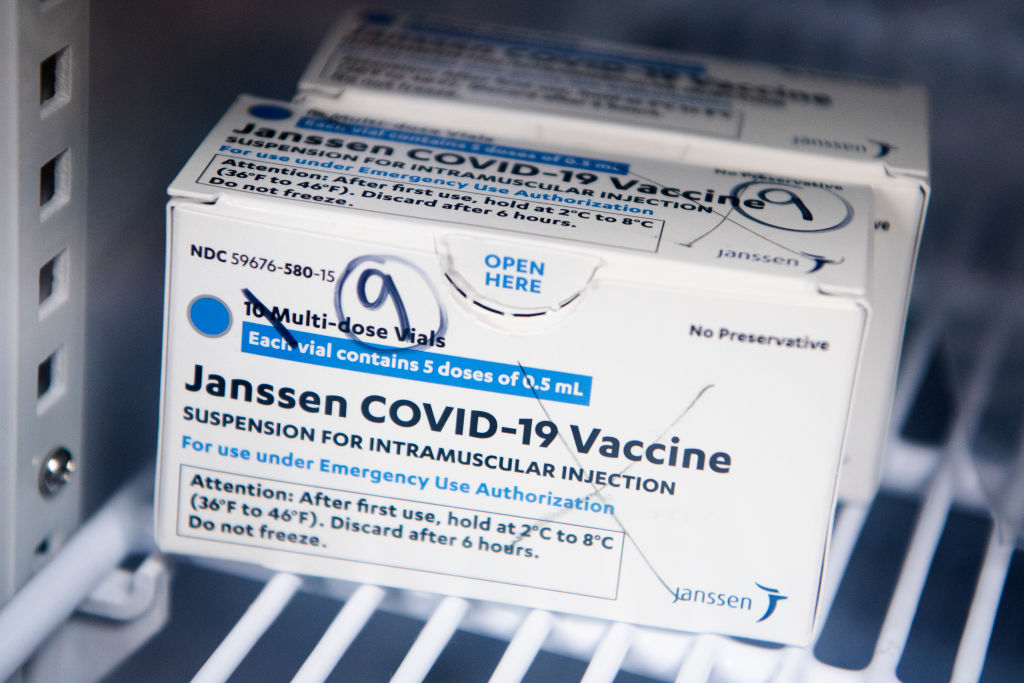
The FDA has announced that the distribution of the Johnson & Johnson COVID-19 will be paused after six women in the U.S. had rare blood-clotting after receiving the vaccine. The women were ranging in ages from 18 to 48, and while six people may not seem like a lot precautions are being taken swiftly. There have been approximately 6.8 million shots given with the majority of people not having issues.
The pausing of the Johnson & Johnson vaccine does not affect the other two vaccines from Pfizer and Moderna. But if you did receive the Johnson & Johnson shot and are having to severe stomach pain, leg cramps, or severe headaches you should call your healthcare provider right away. If you do not know what shot you received, check your CDC vaccination card where it is noted what brand shot you received. If you have an appointment scheduled to receive the Johnson & Johnson COVID-19 vaccine, it is advised to call your provider for further directions.
Get Breaking News & Exclusive Contest in Your Inbox:
[ione_media_gallery id=”3917798″ src=”https://newsone.com/” overlay=”true”]
The Latest:
- After Party Drama at the Oscars!
- Sybil Wilkes Breaks Down What We Need to Know: March 20, 2026
- Guess Who Got the Greenlight for Season 2?!
- Latto Reveals Pregnancy with ‘Big Mama’ Album Cover and New Video
- Tasha K Lists Newly Renovated Home for Sale Amid $4M Cardi B Judgment
- Win Tickets to Floetry!
- Chuck Norris, ‘Walker, Texas Ranger,’ Star Dies At 86
- Kenya Barris Teams Up With Revolt To Launch Revolt Labs
- Tyrese and Tank Set for Verzuz Showdown
- Trump Claim About Epstein Fallout Disproven By 2009 Email








